For the 2021-2022 grant cycle, the University of Arizona Sarver Heart Center awarded six Investigator Awards. These research projects focus on the testing, treatment, and education of the heart.
“Investigator awards have been a pillar of the Sarver Heart Center for years. We are appreciative of all the donors who continue the success this program. This funding is most often awarded to early career investigators and trainees, for innovative pilot projects. We see the return on the investment as the preliminary data collected via the pilot projects frequently lead to large external grant awards,” said
Carol Gregorio, PhD, interim director of the UArizona Sarver Heart Center.
Sarver Heart Center Investigator Awards provide variable amounts of funding per project.
Feasibility of Pressure-Volume Loop Analysis and Invasive Cardiopulmonary Stress Testing via Concomitant Right Heart Catheterization and Cardiac Magnetic Resonance Imaging
 Scientists: Tushar Acharya, MD, MPH, Assistant Professor, Division of Cardiology with Franz Rischard, DO and Nancy Sweitzer, MD, PhD
Scientists: Tushar Acharya, MD, MPH, Assistant Professor, Division of Cardiology with Franz Rischard, DO and Nancy Sweitzer, MD, PhD
Donors: Ella Waltz & Thomas Berresford Heart Research and Phil and Bobby Hanft Young Investigator Award
Dr. Acharya’s study is seeking a more efficient, streamlined method for evaluating the performance of the right side of the heart and lung function in patients with pulmonary hypertension, a serious condition that affects these organs. Right ventricular pressure-volume loops (RV PV-loops) are the gold standard for diagnosing and monitoring pulmonary hypertension and blood flow pressures in the right side of the heart, but are challenging to measure and require expensive and highly specialized equipment with highly trained operators. Without comprehensive evaluation of RV PV-loop derived coupling between the heart and lungs is important in optimal diagnosis and management of pulmonary hypertension. Adding an exercise measure provides an even higher level of functional assessment. However, this gold standard is underutilized clinically because of its cumbersome methodology.
Recently, it has been shown that combining readily available invasive pressure measurements with CMR for RV volume measurement have made assessment more feasible. Unfortunately, at present these measurements cannot be performed simultaneously, which can introduce errors if the patient’s hemodynamic condition changes between tests. Dr. Acharya, a cardiac imaging specialist, will collaborate with Dr. Rischard, a pulmonary hypertension specialist, to evaluate the feasibility of simultaneously performing right heart catheterization and CMR with exercise testing to obtain comprehensive simultaneous RV PV-loops, with and without exercise. The Investigator Award will allow the research team to enroll 10 pulmonary hypertension patients scheduled to undergo a clinically indicated invasive cardiopulmonary exercise test to test this methodology.
Therapeutic Efficacy of ATF6-Activating Compound in a Swine Model of Acute Myocardial Infarction
 Scientists: Erik Blackwood, PhD, postdoctoral fellow, University of Arizona College of Medicine – Phoenix with Christopher Glembotski, PhD and Steven Goldman, MD
Scientists: Erik Blackwood, PhD, postdoctoral fellow, University of Arizona College of Medicine – Phoenix with Christopher Glembotski, PhD and Steven Goldman, MD
Donors: Irving J. Levinson Memorial Research Award and J.G. Murray
Ischemic heart disease, also called coronary artery disease, is the leading cause of deaths worldwide, mainly due to heart attacks which occur when the coronary artery is blocked by cholesterol plaque and blood clots. While working as an EMT after college, Dr. Blackwood kept busy transporting many patients suffering from heart and stroke emergencies. Curious about the causes and progression, he volunteered to work in Dr. Glembotski’s cardiovascular research lab, located in San Diego at the time. The idea of translational research and drug discovery inspired him to pursue his doctoral work at the cardiovascular molecular level.
Heart attack survivors often develop irreparable injury to the heart muscle, leading to eventual heart failure. In prior research, Dr. Blackwood has shown that activation of stress-responsive signaling pathways via a novel small molecule which he and his team discovered provides a promising approach for reducing heart-attack-associated injury and heart-muscle damage. The Investigator Award provides him the opportunity to study toxicity and effectiveness of the identified pharmacological compound in a clinically relevant large animal model. “These proposed studies are significant because they represent crucial next steps in development of this small molecule as a potential therapy prior to moving towards a clinical trial,” said Dr. Blackwood, who plans to use the data generated to support an application for NIH funding.
Since moving with the Glembotski Lab to the College of Medicine – Phoenix in 2021, Dr. Blackwood has found the resources and collaboration between two colleges of medicine to be incredibly supportive of translational research. His research will utilize facilities in both the Goldman Lab at UA Sarver Heart Center in Tucson and the Glembotski Lab in the UA Translational Cardiovascular Research Center in Phoenix.
Machine Learning with Representative Sampling and Bacterial Vitamin K for Prediction of Stable Warfarin Dose
 Scientists: Heidi Steiner, doctoral candidate, Pharmacy Practice and Science with Jason Karnes, PharmD
Scientists: Heidi Steiner, doctoral candidate, Pharmacy Practice and Science with Jason Karnes, PharmD
Donors: The Finley and Florence Brown Endowed Research Award
Focusing new research on an old drug – warfarin - Steiner is studying the utility of artificial intelligence and machine learning techniques, along with vitamin K producing bacteria in the gut in predicting a patient’s therapeutic dose. After more than 50 years in use, warfarin adverse drug events remain a major clinical concern, with approximately 10% of users reporting adverse events. As anyone who has ever used warfarin, or coumadin, knows, the drug requires careful monitoring, and levels can be all over the place. As a potent anticoagulant, having levels of warfarin out of the “target range” can result in excess bleeding or clotting, potentially life-threatening complications. In addition, warfarin interacts with the user’s diet, and patients are frequently prohibited from eating dark green, leafy vegetables (which we of course recommend in all other circumstances! #PlantBasedDiet. Green leafy vegetables contain high levels of Vitamin K, which can increase clotting of blood. Since warfarin is used clinically to thin the blood, anything counteracting that effect can be harmful. Warfarin stable dose algorithms aim to predict a patient’s therapeutic warfarin dose to avoid adverse drug events, but not all populations or factors influencing blood levels are accounted for in current algorithms.
Steiner will apply two state-of-the-art research methods to warfarin dose prediction: machine learning and metagenomics. Machine learning uses artificial intelligence algorithms to identify patterns in and learn from data. Metagenomics will allow Steiner to read the genome of thousands of organisms from the gut simultaneously, thereby surveying the ability of the microbiome to produce vitamin K. “We are investigating the benefit of both machine learning models that incorporate previously excluded Latinx populations, and the composition of the gut microbiome to see if incorporation of these tools improves warfarin dose prediction. Our hope is that by encouraging a warfarin prediction model to learn from diverse populations of data from warfarin users, we can improve prediction for all. We want to give back to our community by addressing the gaps in knowledge regarding warfarin therapeutic dose prediction in a Latinx population, and work to improve the lives of all warfarin users by advancing the research methods utilized in warfarin dose prediction,” said Steiner.
Participants who volunteer for this clinical study are adult warfarin users able to donate stool samples. Visit
here to learn more or check your eligibility.
3-Dimensional MRI Reconstruction of the Heart for Clinical and Teaching Use
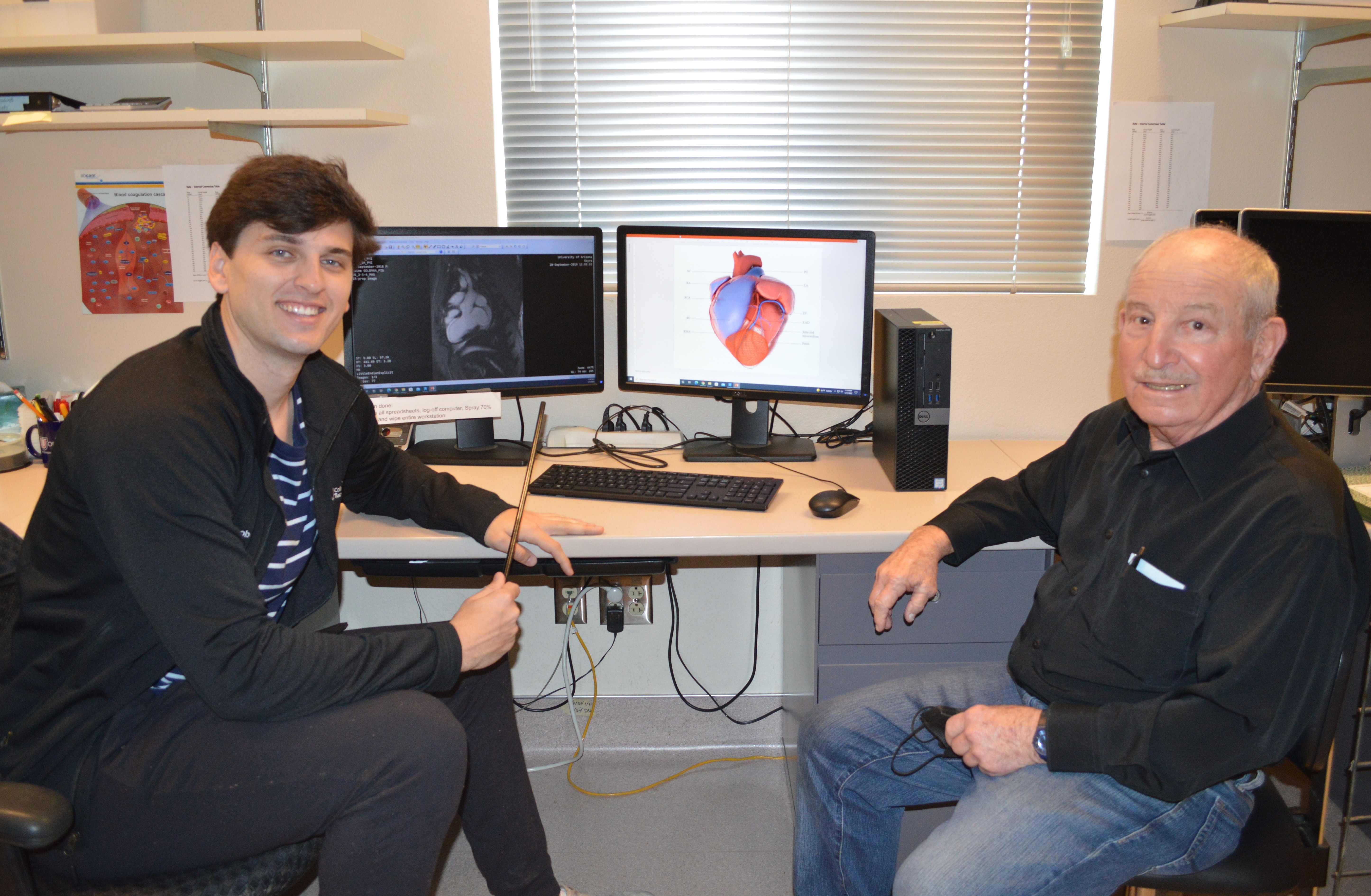 Scientist: Jacob Ref, medical student, Class of 2025, with Steve Goldman, MD
Scientist: Jacob Ref, medical student, Class of 2025, with Steve Goldman, MD
Donors: Margarito Chavez Undergraduate / Medical School Student Awards
Working with scientists in the Goldman Lab, who are developing technology and techniques to repair and regenerate damaged heart muscle, Jacob Ref used his growing knowledge of clinical medicine to observe that the 2-dimensional black and white radiology images were perhaps not the best tool to provide a true scope of the damaged area to guide research procedures. To bridge the translational gap from science to the patient, a better visual aid will help scientists precisely locate the site of damage and better place cell-based therapeutics precisely in areas of heart damage. Jacob has been working alongside Brad Baxley, a local architect and creative director at Part to Whole, along with experts in cardiovascular imaging, to execute a methodology that creates a 3D model of the heart that is anatomically correct based on clinical and research images.
A better visual aid will also improve education to those learning the vascular and myocardial effects of ischemia. Educational benefits of a 3-dimensionally architected heart will not only help patients visualize what a heart attack means in terms of damage to the heart, but also help to teach the next generation of healthcare workers how to appreciate the extent of ischemic damage to the heart.
Now, a first-year medical student, Jacob started working in the Goldman Lab when he was an undergraduate in physiology.
Novel Research Project Awards in the area of Cardiovascular Disease and Medicine
 Prohibitin as a Novel Therapeutic Target for Cardiac Fibrosis
Prohibitin as a Novel Therapeutic Target for Cardiac Fibrosis
 Scientists: Alexandra (Bobbie) Garvin, PhD, postdoctoral fellow, physiology, Basic Medical Sciences, College of Medicine – Phoenix with Taben Hale, PhD
Scientists: Alexandra (Bobbie) Garvin, PhD, postdoctoral fellow, physiology, Basic Medical Sciences, College of Medicine – Phoenix with Taben Hale, PhD
Donors: Dr. Alex Frazer and Frank H. Frazer, Robert C. and Olive M. Johnson, John T. and Janet K. Billington
Heart muscle injury, often caused by poorly controlled high blood pressure or heart attacks, leads to activation of cardiac fibroblasts. These cells produce the matrix that makes up scar tissue. Prolonged activation of these cells leads to cardiac fibrosis, resulting in heart muscle stiffening that impedes the heart’s ability to pump blood efficiently. This can progress to heart failure and can also increase the risk for sudden cardiac death. Currently, no interventions are available clinically to stop or reverse cardiac fibrosis.
Dr. Garvin’s research aims to identify the role of a novel molecular target for “turning off” activated cardiac fibroblasts. “I came to the Hale Lab to broaden my horizons in cardiac physiology. While a graduate student at Penn State, my cardiovascular research focused on preventing the loss of heart muscle cells after a heart attack,” said Dr. Garvin. “Mitochondria are the cellular power source and can determine whether a cell lives or dies. My prior research on targeting mitochondria to protect contractile heart muscle cells has fueled this new direction in cardiac fibroblasts.” Now, Dr. Garvin is studying ways to leverage mitochondrial-mediated cell death as a force for good by curtailing the activated cardiac fibroblasts that cause fibrosis.
“The study of energetics in cardiac fibroblasts is a unique research direction,” said Dr. Garvin, who is working to obtain findings that lead to national funding of her own research program, using this investigator award to move toward independence in her scientific career. Her research aims to lead to cell-specific gene therapy for patients with heart failure.
Probiotic treatment to reverse menopausal intestinal epithelial changes in organoids and reduce myocardial infarct size
 Scientists: Vito Marino, PhD student, Physiological Sciences, with John Konhilas, PhD
Scientists: Vito Marino, PhD student, Physiological Sciences, with John Konhilas, PhD
Donors: Ralph and Shirley Morgan Cardiovascular Research Award and J.G. Murray
Cardiovascular disease is one of the leading causes of death worldwide. Although the incidence in women is usually lower than men, women have a higher mortality and worst prognosis after acute cardiovascular events. These discrepancies are likely due to the loss of the sex hormone estrogen during menopause, a condition almost all women will experience. Estrogen plays many physiological roles in the body that are only beginning to be understood. Of note, estrogen appears to be involved in the normal interplay between healthy microbes living in the gut and cells important for maintaining a barrier along the gastrointestinal tract. The goal of this project is to find the connection between loss of estrogen during menopause and changes in gut microbes and gut permeability that contribute to worsening cardiovascular disease risk and outcomes after menopause in women.
“We’ve learned that we can’t just give back estrogen without health risks. The body does continue to produce estrogen beyond menopause; however the amount and types of estrogen change,” said Dr. Konhilas. This work will provide a novel paradigm targeting the gut microbiome with novel therapies in an attempt to reduce cardiovascular risk.
“By studying bacteria collected in the gut, we know that 350 genes are important to estrogen regulation,” said Marino, who previously studied an enzyme used to inhibit estrogen receptors in breast cancer. His question now: can we use this to increase natural estrogen? “Cells line the gut forming a protective wall and menopause creates chips in the wall that allow the transport of pathogens to pass through, increasing inflammation, cardiovascular disease, obesity and metabolic syndrome. Menopause takes the foot off the brakes of this protective system. Can we restore the brakes?”
 Scientists: Tushar Acharya, MD, MPH, Assistant Professor, Division of Cardiology with Franz Rischard, DO and Nancy Sweitzer, MD, PhD
Scientists: Tushar Acharya, MD, MPH, Assistant Professor, Division of Cardiology with Franz Rischard, DO and Nancy Sweitzer, MD, PhD Scientists: Erik Blackwood, PhD, postdoctoral fellow, University of Arizona College of Medicine – Phoenix with Christopher Glembotski, PhD and Steven Goldman, MD
Scientists: Erik Blackwood, PhD, postdoctoral fellow, University of Arizona College of Medicine – Phoenix with Christopher Glembotski, PhD and Steven Goldman, MD Scientists: Heidi Steiner, doctoral candidate, Pharmacy Practice and Science with Jason Karnes, PharmD
Scientists: Heidi Steiner, doctoral candidate, Pharmacy Practice and Science with Jason Karnes, PharmD  Scientist: Jacob Ref, medical student, Class of 2025, with Steve Goldman, MD
Scientist: Jacob Ref, medical student, Class of 2025, with Steve Goldman, MD Prohibitin as a Novel Therapeutic Target for Cardiac Fibrosis
Prohibitin as a Novel Therapeutic Target for Cardiac Fibrosis Scientists: Alexandra (Bobbie) Garvin, PhD, postdoctoral fellow, physiology, Basic Medical Sciences, College of Medicine – Phoenix with Taben Hale, PhD
Scientists: Alexandra (Bobbie) Garvin, PhD, postdoctoral fellow, physiology, Basic Medical Sciences, College of Medicine – Phoenix with Taben Hale, PhD Scientists: Vito Marino, PhD student, Physiological Sciences, with John Konhilas, PhD
Scientists: Vito Marino, PhD student, Physiological Sciences, with John Konhilas, PhD
